StressMarq’s Active Tau Pre-Formed Fibrils (PFFs)
StressMarq is excited to offer active tau proteins to help researchers study tau aggregation, a hallmark of neurodegenerative diseases including Alzheimer’s. StressMarq is the first to offer active tau preformed fibrils (PFFs) and filaments for neuroscience research. The process of tau aggregation can be seeded by active tau PFFs, which recruit monomers to form larger tau fibrils. This has been demonstrated in thioflavin T assays where an increase in fluorescence, indicative of tau fibrillization, is seen when active tau PFFs are combined with active tau monomers. Certain tau PFFs have been injected into P301L mice, where they seed tau aggregation and induce tau pathology in the hippocampus.
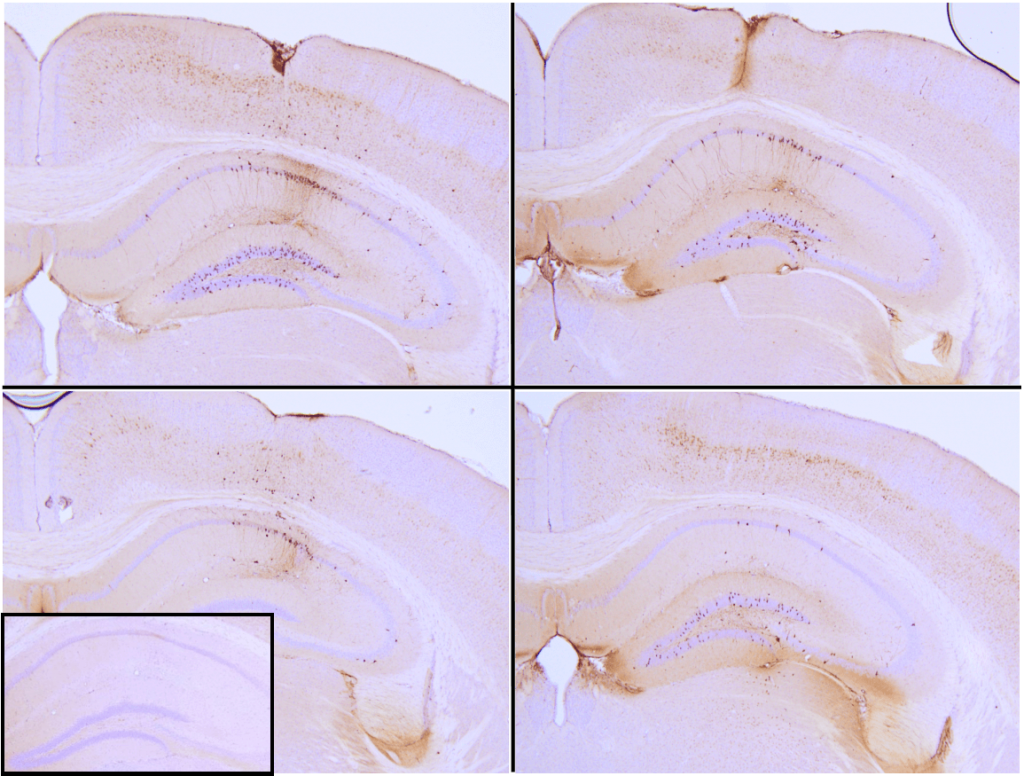
Immunohistochemistry analysis of P301L mouse hippocampus injected with K18 P301L tau PFFs (SPR-330) shows seeding of tau pathology at injection site nine weeks post-injection. AT8 (pSer202/pThr205) tau antibody shows tangle-like inclusions. Inset: negative control. Experiments performed at reMYND N.V.
Monomers and fibrils are available both in the full-length isoform of the tau protein (2N4R or Tau-441) or a truncated form (K18). Tau-441 has a molecular weight of approximately 46 kDa, whereas K18 tau has a molecular weight of approximately 15 kDa. dGAE is a fragment of the tau protein consisting of amino acids 297-391. It is one of the core PHF subunits,1 includes both microtubule-binding domains and proline-rich regions, and assembles into PHF-like fibrils in vitro without additives or templates.2
Proteins are available as wild-type or with a variety of mutations. P301S and P301L mutations occur in exon 10 and are associated with frontotemporal dementia. The P301S mutation reduces tau’s ability to assemble microtubules, and the P301L mutation promotes beta-sheet formation and the formation of PHFs. Both P301S and P301L mutant transgenic mouse models are used in tau research. The K280 deletion mutation is also associated with frontotemporal dementia and promotes fibrillization into paired helical filaments (PHFs) in the absence of heparin and other inducers. The C322A mutation also increases tau’s ability to form PHFs.2
PFFs induce tau aggregation; full-length PFFs may be more effective in seeding fibrillization, but a combination of both can be particularly toxic to neurons. Most tau varieties are fibrillized using a heparin scaffold; soluble tau filaments are fibrillized using a linear anionic scaffold. dGAE tau and K18 K280 deletion tau both fibrillize without scaffolds.
Tau PFFs can be expressed in baculovirus in addition to E. coli. Baculovirus tau PFFs are post-translationally modified, fibrillize without a scaffold, and are endotoxin-free*.
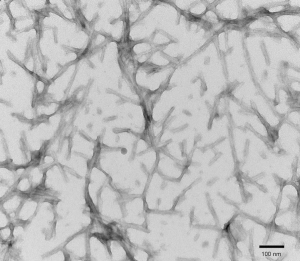
TEM of active recombinant Tau441 (2N4R), P301S mutant preformed fibrils (PFFs) (SPR-329).
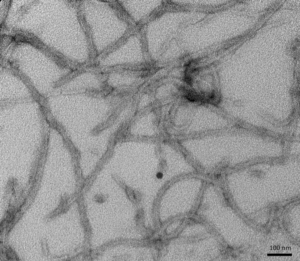
TEM of active recombinant Tau (K18), P301L mutant preformed fibrils (PFFs) (SPR-330).
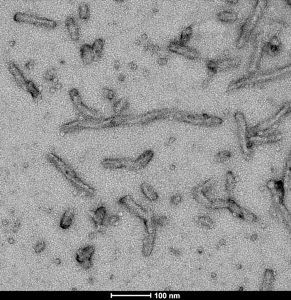
TEM of Human Recombinant Tau Protein Filaments (SPR-463)
1. Novak, M. et al. (1993) EMBO J. 12, 365-370.
2. Al-Hilaly, Y.K. et al. (2017) J. Mol. Biol. 429(23):3650-3665.
*. Endotoxin <0.05 EU/mL (below the detectable range of the assay)
StressMarq is in the process of developing new types of fibrils for neurodegenerative disease research as well as thoroughly characterizing our current products. We currently have several types of tau PFFs and filaments available. This table summarizes the differences between them:
| Catalog No. |
Protein |
Species |
Expression System |
Length/Fragment |
Mutant |
Monomer source |
Fibrillization Scaffold |
In vivo activity |
| SPR-329 |
2N4R, P301S PFFs |
Human |
E. coli |
2N4R (441-amino acid) Full length |
P301S |
SPR-327 |
Heparin |
Under Investigation |
| SPR-480 |
2N4R PFFs |
Human |
E. coli |
2N4R (441-amino acid) Full length |
WT |
SPR-479 |
Heparin |
Under Investigation |
| SPR-329-A488 |
ATTO 488-conjugated 2N4R, P301S PFFs |
Human |
E. coli |
2N4R (441-amino acid) Full length |
P301S |
SPR-327 |
Heparin |
Under Investigation |
| SPR-330 |
K18, P301L PFFs |
Human |
E. coli |
K18 (4R) Truncated |
P301L |
SPR-328 |
Heparin |
Induces tau phosphorylation and aggregation in P301L transgenic mouse hippocampus |
| SPR-461 |
dGAE PFFs |
Human |
E. coli |
dGAE (AA297-391) Truncated |
WT |
SPR-444 |
|
Under Investigation |
| SPR-462 |
dGAE, C322A PFFs |
Human |
E. coli |
dGAE (AA297-391) Truncated |
C322A |
SPR-445 |
|
Under Investigation |
| SPR-463 |
2N4R, P301S Filaments |
Human |
E. coli |
2N4R (441-amino acid) Full Length |
P301S |
SPR-327 |
Linear Anionic Scaffold |
Under Investigation |
| SPR-471 |
2N4R, P301S PFFs |
Human |
Baculovirus |
2N4R (441-amino acid) Full length |
P301S |
SPR-473 |
|
Under Investigation |
| SPR-475 |
2N4R, P301S PFFs |
Mouse |
E. coli |
2N4R (441-amino acid) Full Length |
P301S |
SPR-474 |
Heparin |
Under Investigation |
| SPR-477 |
K18, K280 Deletion PFFs |
Human |
E. coli |
K18 (4R) Truncated |
ΔK280 |
SPR-476 |
|
Under Investigation |
Tau Publications
Astrocyte senescence and SASP in neurodegeneration: tau joins the loop
- Ungerleider, K. et al. (2021) Cell Cycle. 1-13.
- Read more on our blog.
Aberrant AZIN2 and polyamine metabolism precipitates tau neuropathology
- Sandusky-Beltran, L.A. et al. (2021) J Clin Invest. 131(4):e126299.
- Read more on our blog.
Cornel Iridoid Glycoside Regulates Modification of Tau and Alleviates Synaptic Abnormalities in Aged P301S Mice
- Yang, Cc. et al. (2020) Curr Med Sci. 40, 1040–1046(2020).
- Read more on our blog.
Tau Protocols
Handling Instructions – Learn how to handling tau proteins safely and effectively
Thioflavin T Assay Protocol – Thioflavin T is a dye that binds to beta-sheets and allows the for the study of tau aggregation.
Learn about the best storage conditions and experimental methods for PFFs.
Tau Protein Resources
Types of Tau Fibrillar Constructs – Learn about our tau fibrillar constructs and their characteristics.
Which Tau Construct Should I Use? – A Guide to Modelling Tauopathies
StressMarq Tau Research Tools Poster – A summary of proteins and antibodies available for tau research.
Studying Alzheimer’s and Parkinson’s Disease using Pre-Formed Fibrils – An interview with StressMarq President & CEO Ariel Louwrier about PFFs in neurodegeneration research.
Modelling Alzheimer’s Disease in Cells and Animals – Read about different models used in AD research and their strengths and weaknesses.
Tau Mutations and their role in Neurodegenerative Disease – Learn about the role of various tau mutations in aggregation and disease progression.
Tau Phosphorylation in Alzheimer’s Disease – Learn how phosphorylation at certain sites is linked to tau aggregation.
Tau Post-Translational Modifications: An Overview of the Baculovirus Expression Vector System – What are the advantages of the baculovirus expression vector system in recombinant protein production?
Tau Post-Translational Modifications in Alzheimer’s Disease: Diagnostic and Therapeutic Opportunities – Learn about the role of tau post-translational modifications in AD and potential therapeutic strategies.